Florence Luyten, Account Manager at Indaver for precious metal recycling from liquids, explains how advances in recycling technologies are enabling a circular economy for precious metals used as homogenous catalysts.
Why recycle?
Homogeneous catalysts lie at the heart of industrial processes producing many pharmaceutical and agrochemical ingredients, or other fine chemicals. They include acetates, chlorides, nitrates, oxides or sulphates of precious metals such as palladium (Pd), rhodium (Rh), platinum (Pt), ruthenium (Ru), iridium (Ir) and silver (Ag). By lowering energy barriers, homogeneous catalysts drive chemical processes in global industries, leading an unending, unyielding demand for their component precious metals.
Conventionally, precious metals have been mined from natural mineral deposits. However, the rapid depletion of these limited resources, as well as the environmental impact of excavation, make mining unsustainable. The long-term ecological effects of mining can persist for decades, with derelict lands permanently altering the landscape, and rendering large areas of land unusable for agriculture or habitation.
Fortunately, there is a ready alternative. Precious metals are ideal candidates for a circular economy[1]. Although the recovery process can be challenging, because it involves recovering low concentrations of precious metals from large volumes of liquids, the high value and scarcity of precious metals are widely considered to make a circular approach worthwhile.
Testament to this belief, the past few years have seen a significant increase in the recycling of precious metals, contributing to their sustainability and reducing the environmental impact of sourcing them through primary mining. For palladium, for example, we have clearly demonstrated that concentrations as low as 20 ppm (and as high as 6,000 ppm) are economic to recycle from liquid streams. A 2025 study performed by Indaver in collaboration with KU Leuven, a well-known research university located in Leuven, Belgium, has shown that Indaver’s recycling processes for precious metals resulted in carbon savings of up to 98%, compared with primary mining.
Recycling precious metals
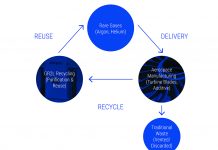
Advances in recycling processes are transforming the precious metals industry towards more sustainable approaches. Indaver has developed a strong offering in the recycling of precious metals such as palladium, rhodium, platinum, ruthenium, iridium and silver. The company’s facility in Antwerp, Belgium, specialises in recovering these precious metals directly from the liquid phases originating from processes based on homogeneous precious metal catalysts, primarily from the agrochemical, speciality chemical and pharmaceutical industries.
Techniques like precipitation, adsorption and thermal methods enable the recovery of precious metals from liquids streams in every compound or shape. Based on laboratory testing, the recovery method that offers the highest efficiency at the most favourable cost is selected, enabling a tailored processes for precious metal isolation from hazardous streams (e.g. toxic, low pH, mixtures). For example, some streams might be suited to both precipitation and adsorption, but the adsorption material might be very expensive, or it might become quickly saturated; in this case, precipitation would be chosen.
There are two installations at the Antwerp site: Line 1 uses thermal methods, while Line 2 performs precipitation and adsorption. Typically, organic and solvent streams are routed to Line 1, while aqueous streams and streams with sediment are routed to Line 2 (Figure 1). This two-line approach enables flexibility in providing the optimal process for any individual stream, while ensuring a completely pure process that is dedicated to precious metals recovery and allows tailored streams to achieve high efficiencies. Case Studies 1 and 2 provide two recent examples of palladium extraction from organic waste and organic aqueous streams, respectively. Waste streams are never mixed to avoid contamination – the work is done in a batch process with full traceability.
Case study 1:
To recover palladium from an organic waste stream including 600 ppm palladium.
_____________________________________________________
Precious metal extraction:
Thermal reduction on the Antwerp site’s Line 1
=> ~5% metal fraction
(semi-solid with high precious metal concentration)
Thermal treatment of metal fraction
=> 0.1–0.3% volume of platinum-group metal (PGM) rich ashes with ~30% palladium
______________________________________________________
Liquid fraction treatment:
94–96% liquid fraction; solvent with minor impurities
Solvents were recycled
=> full circular process for the entire waste stream
Case study 2:
To recover palladium from an organic aqueous mixture including 30 ppm palladium.
_____________________________________________________
Precious metal extraction:
Treatment on Antwerp site’s Line 2
=> 2–5% metal fraction
(dry powder with high precious metal concentration)
Thermal treatment of metal fraction
=> 1–3% volume of platinum-group metal (PGM) rich ashes
______________________________________________________
Liquid fraction treatment:
95–98% liquid fraction; aqueous solvent
Treated on the on-site waste to energy installation
=> maximising energy recovery and safe destruction of hazardous components
Liquid fraction treatment
Once the precious metal has been recovered, a liquid fraction remains that also needs to be treated. This includes liquids that may be characterised as hazardous, toxic, flammable or corrosive. It is very important that any installations recovering precious metals also have the ability to deal with these liquid fractions, which may make up 95% or more the liquid stream, by volume (see Case Studies 1 and 2). Appropriate consideration for the liquid fraction ensures that the precious metals can be recovered efficiently, while any remaining hazardous components are treated using the most suitable and efficient techniques.
While hazardous components are safely destroyed, valuable solvents can be further purified and reused. Once the metal fraction has been removed, in the case of solvent streams, developments in available technologies enable the efficient and sustainable of solvent recycling processes[2]. Companies can typically expect to recover up to 95% of used solvents, even from more complex mixtures. In terms of quality and purity, recovered solvents are equal to virgin material.
Conclusion
It is both ecologically and economically beneficial to recover precious metals and re-use them as homogenous catalysts. Using this approach, industrial manufacturers have a constant and sustainable supply of precious metals, a simplified, localised supply chain, and a reduced carbon footprint to contribute to their sustainability objectives. The recovery and recycling of precious metals and solvents improves process efficiency, saves costs and simplifies the supply chain by eliminating the need to constantly purchase new materials. Added to this, it brings considerable environmental benefits as it reduces the need for polluting mining activities and demonstrates up to 98% reduction in carbon footprint.
Indaver is dedicated to extracting precious metals and solvents from liquid streams while ensuring the safe destruction of hazardous components. Developing and applying tailored processes – such as precipitation, adsorption and thermal methods – for precious metal isolation from hazardous solvent or aqueous based streams enables the efficient recovery of precious metals, even at very low ppm concentrations. By driving advances in recycling technology and the increasing demand for recycled materials, we are closing the loop on precious metals and clearing the path towards a circular economy for homogenous catalysts.
indaver.com
References
Yao C, Hengjun S, Yuqing R, et al. Systematic assessment of precious metal recovery to improve environmental and resource protection. ACS ES&T Engineering 2022;2:1039–1052.
Barker S. High-End Circular Solvents: A Path to Sustainability. Chemicals Industry Journal 2025;38:42–43.