Companies who pre-registered substances for KKDIK Compliance in Turkey, are living a reflection of the year 2009 – first quarter after the REACH pre-registration deadline of 2008 was over.
The initial MBDF communications are not yet as intense as the first quarter of 2009 experienced in the EU but there are still solid approaches from some dedicated Lead registrant candidates who are mostly data holders. EU REACH allowed companies to register in a 10 years-time period while KKDIK gives 3 years for all companies to register after the pre-registration deadline of 31st December 2020. Therefore, it is anticipated that the communications will speed up considerably in the next coming months.
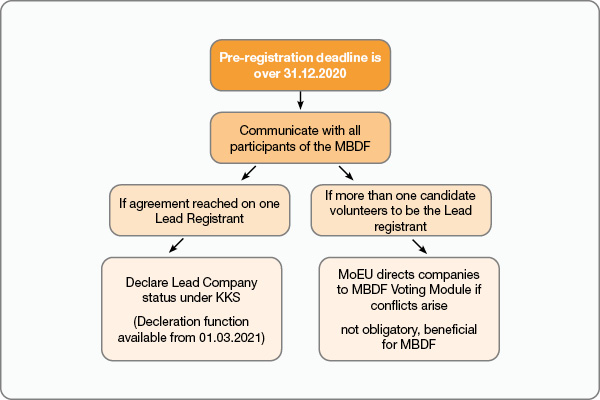
Although MBDF communications have slowly been started even before January 2021 by some pre-registrant companies, the Turkish Ministry of Environment and Urbanisation (MoEU) took control of the predicted future disagreements within the MBDF by scheduling introduction of additional functions and integration of new modules to the online Chemicals Registration System (KKS).
The MoEU will activate “Lead Company Declaration” function under KKS on 1st March 2021. This function will be simultaneously accompanied by integration of a voting module that will allow companies to vote for the lead company of their choice, if there is more than one candidate to avoid any possible conflicts. Although it is not mandatory to vote for the Lead Registrant, the MoEU may request MBDF participants to vote using the module under KKS or refer to the results of the voting, if deemed necessary. One Leading company will be assigned for each reference substance.
The MoEU also emphasizes the importance of communication with all the KKDIK MBDF participants before any company declares its decision as a Lead company under KKS with the new functionality that will be active on 1st March. (Ref: MoEU announcement made on 15th February 2021 on the Ministry’s official website)
If there is a conflict while choosing the Lead for a substance within the MBDF, it is advised to get in contact with the MoEU. In such a case, MBDF participants will be informed and directed to voting module to make their decisions and determine the lead registrant.
COMPANY SIZE MATTERS
Meanwhile, company size has always been an issue in EU REACH registrations especially as wrong company size declarations were inspected by European Chemicals Agency. This let to payment of incorrect registration fees which is usually the registration fee specified for the smaller company size. Eventually, administrative charges and additional payments to balance out the accurate registration fee were requested by ECHA.
In order to prevent any misunderstandings, fee calculator functionality has also been integrated into KKS. The fees to be paid according to company size, tonnage band and registration type can be calculated and a specified fee appears at the registration dossier submission stage. The conditions and documents required for Turkish companies and local Only Representatives appointed by non-Turkish companies to benefit from the reduced fees for SMEs are also summarised and clarified by the MoEU via the latest announcement on the Ministry website.
While the necessity of establishing a consortium in Turkey is still not clear for some substances or substance groups, it is crystal-clear that the data sets for KKDIK registrations already exists in the control of EU consortia. This applies to substances that are already registered to ECHA under EU REACH. Many consortia members in the EU are happy to share the right to use the data to comply with regulations in the other regions. The use of data for UK-REACH compliance has been one of the discussions in several EU consortia while Turkey is certainly one of the regions under discussion. Hopefully, this will decrease the amount of time spent and cost to get the dossiers ready for submission without the need for extra testing.
RGS Group based in Brussels, Belgium with offices in Istanbul, Turkey is representing global companies located in over 50 countries as a local Only representative through its Turkish presence. As a leading company, RGS also transfers 13 years of regulatory experience predominantly in EU REACH experience to its operations in Turkey. Do not hesitate to contact RGS, if you need more details on our services. We can assess your compliance status and build tailor-made solutions for your company.
Dr.Yaprak Yüzak Küçükvar
REACH Global Services Group
Director, Global Regulatory Affairs & Product Stewardship